Key Aspects of MDR and Patient Safety in the Procurement of Medical Devices
Since 2021, the European regulatory framework MDR (Medical Device Regulation) has been closely adhered to in the field of medical technology. MDR has the overarching goal of ensuring patient safety by establishing and imposing strict requirements for transparency, documentation, traceability, and follow-up related to the use of medical technology products.
How does MDR relate to the Procurement of Medical Technology Products?
There are several standards harmonized with MDR that every manufacturer with CE marking on their products is expected to follow. These standards include requirements for biological evaluation, quality management systems, risk management, labeling, and electrical safety.
What MDR clearly emphasizes is the requirement for clinical evidence. This means that every manufacturer must account for their clinical claims with supporting clinical documents and facts. As per MDR, attributing characteristics to a medical technology product without supporting evidence is strictly prohibited. For example, if a medical technology product claims to alleviate pain, there must be clinical evidence not only confirming that the product is intended for this purpose but also demonstrating its effective and safe performance.
According to the MDR regulation, clinical evidence is defined as: “the clinical data and results from clinical evaluations relating to a specific product. Data enabling a precise assessment of the product’s safety and its ability to achieve the intended clinical benefits when used in accordance with the manufacturer’s instructions” (1).

Clinical Evidence in Procurement
To ensure patient safety, it is crucial that procuring authorities, in the evaluation of medical technology products, have a clear understanding of the various types of evidence supporting the manufacturer’s claims about their products. A clinical claim regarding a medical technology product must be substantiated with clinical data, which means evidence based on the product’s use over a specified period in its intended user environment and with its intended users. This is necessary to ensure that the product meets its claimed functions and is safe to use in practice.
Levels of Evidence
Evidence is classified based on the type of clinical data and its quality. Appendix III in MDCG 2020-06 proposes a hierarchy of clinical data to demonstrate compliance with the general safety and performance requirements according to MDR. The highest rank in this evidence hierarchy is held by high-quality clinical studies covering all product variants, including all indications, patient groups, treatment durations, etc., as intended by the manufacturer. Following that are high-quality clinical studies where limitations are made to a specific purpose or a specific patient group. Next in line are high-quality clinical data from, for example, registry studies, followed by outcomes of clinical studies with limitations in methodology.
Supporting evidence for clinical claims can also be backed up by equivalent products where equivalence is demonstrated and meets MDR requirements but is insufficient as clinical data alone to assess compliance with the general performance and safety requirements of the product. Safety monitoring, complaints, and proactive post-market follow-up in the form of surveys are classified as longer levels of clinical data.
At the bottom of the hierarchy, we find technical tests (such as controlled tests on mattresses solely measuring pressure distribution) and laboratory studies conducted under controlled laboratory conditions. These tests can provide supporting evidence and address clinical outcome measures, but according to MDR, they are inherently insufficient as individual evidence to assess the clinical performance to ensure that the products are safe to use in practice (2, 3).
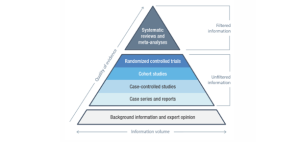
In conclusion, to ensure compliance with the regulations and thus patient safety, it is crucial to demand clinical evidence for medical technology products under consideration for procurement. The ability to distinguish between high-quality clinical data and supporting data is central to guaranteeing that products used in healthcare meet the stringent requirements for patient safety and quality prescribed by MDR.
References:
1. MDR 2017/745, Article 2. Retrieved 2023-12-01. https://eurlex.europa.eu/legalontent/EN/TXT/HTML/?uri=CELEX:32017R0745&from=IT
2. MDCG 2020-06, Regulation (EU) 2017/745: Clinical evidence needed for medical devices previously CE marked under Directives 93/42/EEC or 90/385/EEC A guide for manufacturers and notified bodies. Retrieved 2023-12-01. https://health.ec.europa.eu/system/files/2020-09/md_mdcg_2020_6_guidance_sufficient_clinical_evidence_en_0.pdf
3. Openmd. (2021) Levels of Evidence. Retrieved 2023-09-23. https://openmd.com/guide/levels-of-evidence
